| Taxon names | Citations | Turn highlighting On/Off |







(C) 2013 Sven G. Nilsson. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Citation: Nilsson SG, Franzén M, Pettersson LB (2013) Land-use changes, farm management and the decline of butterflies associated with semi-natural grasslands in southern Sweden. Nature Conservation 18: 31–48. doi: 10.3897/natureconservation.6.5205
Currently, we are experiencing biodiversity loss on different spatial scales. One of the best studied taxonomic groups in decline is the butterflies. Here, we review evidence for such declines using five systematic studies from southern Sweden that compare old butterfly surveys with the current situation. Additionally, we provide data on butterfly and burnet moth extinctions in the region’s counties. In some local areas, half of the butterfly fauna has been lost during the last 60–100 years. In terms of extinctions, counties have lost 2-10 butterfly and burnet moth species. Land use has changed markedly with key butterfly habitats such as hay meadows disappearing at alarming rates. Grazed, mixed open woodlands have been transformed into dense coniferous forests and clear-cuts, and domestic grazers have been relocated from woodlands to arable fields and semi-natural grasslands. Ley has increased rapidly and is used for bale silage repeatedly during the season. Overall, the changed and intensified land use has markedly reduced the availability of nectar resources in the landscape. Species that decline in Sweden are strongly decreasing or already extinct in other parts of Europe. Many typical grassland species that were numerous in former times have declined severely; among those Hesperia comma, Lycaena virgaureae, Lycaena hippothoe, Argynnis adippe, and Polyommatus semiargus. Also, species associated with open woodlands and wetlands such as, Colias palaeno, Boloria euphrosyne and the glade-inhabiting Leptidea sinapis have all decreased markedly. Current management practise and EU Common Agricultural Policy rules favour intensive grazing on the remaining semi-natural grasslands, with strong negative effects on butterfly diversity. Abandoned grasslands are very common in less productive areas of southern Sweden and these habitats may soon become forests. There is an urgent need for immediate action to preserve unfertilized, mown and lightly grazed grasslands. It is also crucial to encourage that management of abandoned grasslands resumes before it is too late. In order to mitigate risks of further species loss and to work towards recovery of threatened butterfly populations using best known practises, we recommend twelve types of management measures favourable for many butterflies.
Land management, conservation, agroecology, semi-natural grasslands, management recommendations, butterflies, Sweden
During the last 100 years, agriculture has experienced profound changes in Europe. In particular, it has become increasingly mechanized because of the pressure for higher yields and the rising price of labour (
Most of southern Sweden is characterised by a forest-dominated landscape, with small-scaled farmland covering about 5 % (
Open habitats associated with a rich butterfly fauna in southern Sweden: A Taxås nature reserve in Kronoberg County, Småland, a pasture with one part experiencing late grazing B An abandoned pasture in Blekinge C A recently abandoned meadow in Småland. Photos: Markus Franzén.
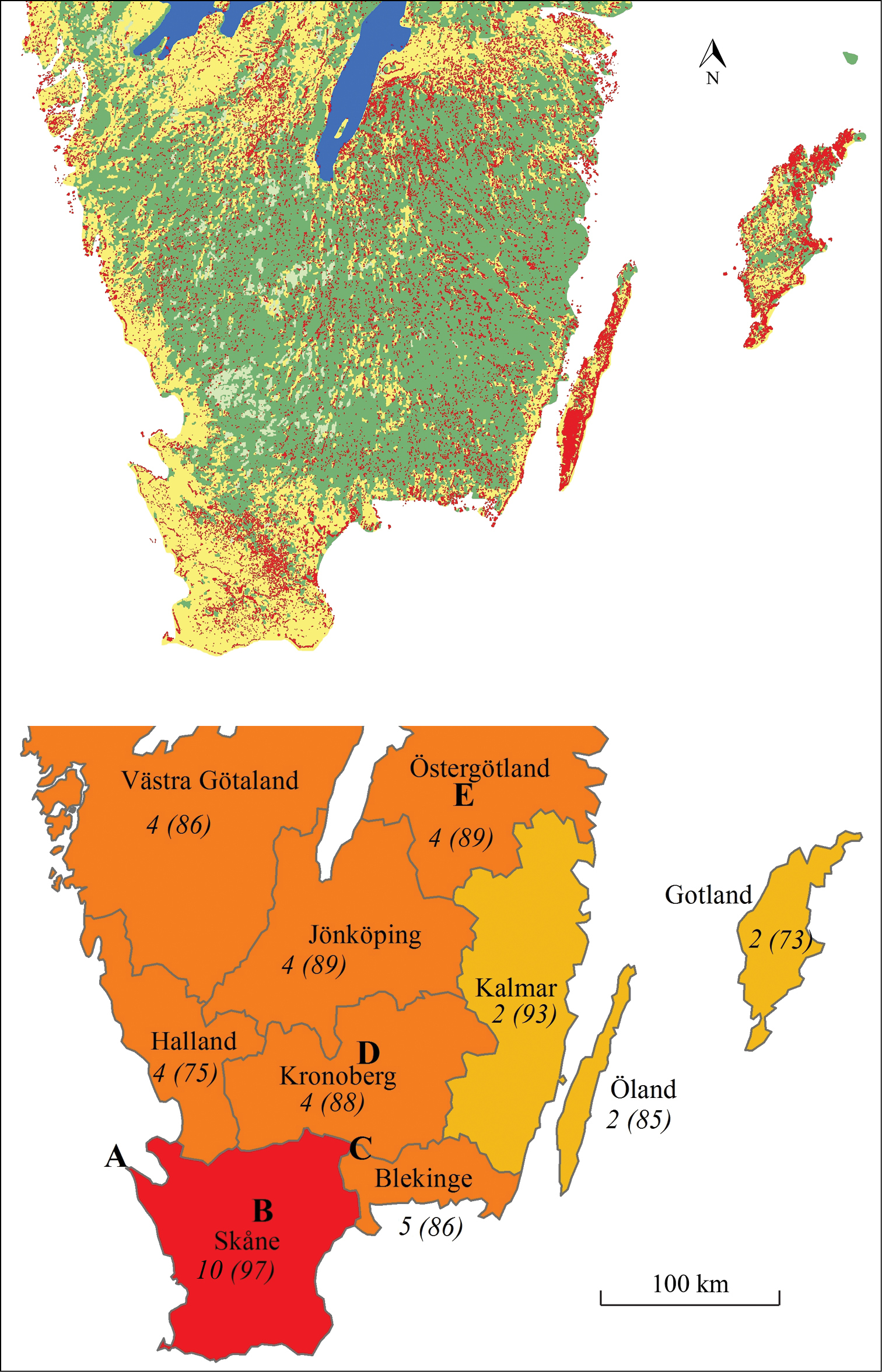
Administratively, the studied area is split into nine counties (Figure 2) which, together with the Baltic island Öland, form the basis for our analysis of extinction events (cf.
Upper map: Main habitats in southern Sweden (dark green= forest; light green = wetlands; yellow = arable fields; blue = water; red = semi natural grasslands; data from (
The proportion of the landscape that consists of pastures and hay meadows in this area is approximately 5% (Data from the Swedish Board of Agriculture;
Five studies during the last decade have documented distinct decreases in butterfly and burnet moth species richness in southern Sweden (Table 1, Figure 2). It should be noted that burnet moths are generally included in butterfly surveys in Sweden because of the two groups’ similar habitat requirements and ecology and we follow this tradition here (
Butterfly and burnet moth species that have disappeared (Extinct), decreased in numbers (Decrease), increased (Increase) or remained relatively unchanged (Unchanged) in abundance in three systematic resurveys in southern Sweden at Kullaberg (Kullaberg;
| Species | Grassland, Wetland or Forest | Kullaberg | Nöbbele | Pastures | Major larval food plants |
|---|---|---|---|---|---|
| Adscita statices* | G | Unchanged | Decrease | Decrease | Rumex acetosella, Rumex acetosa |
| Anthocharis cardamines | G | Unchanged | Decrease | Decrease | Brassicaceae |
| Aporia crataegi | G | Extinct | Extinct | Extinct | Sorbus aucuparia, Crataegus |
| Araschnia levana | G | Increase | - | Increase | Urtica dioica |
| Argynnis adippe | G | Extinct | Extinct | Decrease | Viola spp. |
| Argynnis aglaja | G | Extinct | Unchanged | Decrease | Viola spp. |
| Boloria euphrosyne | W | Extinct | Decrease | Decrease | Vaccinium uliginosum |
| Boloria selene | G | Extinct | Unchanged | Decrease | Viola, especially Viola palustris |
| Coenonympha pamphilus | G | Unchanged | Decrease | Decrease | Poaceae |
| Colias palaeno | W | Extinct | Extinct | - | Vaccinium uliginosum |
| Cupido minimus | G | Extinct | Extinct | - | Anthyllis vulneraria |
| Erynnis tages | G | Extinct | - | Extinct | Lotus corniculatus |
| Favonius quercus | G | Unchanged | Decrease | Decrease | Quercus |
| Hesperia comma | G | Extinct | Extinct | Decrease | Festuca ovina, Festuca rubra, Agrostis vinealis |
| Lasiommata maera | G | Extinct | Decrease | Decrease | Poaceae |
| Leptidea sinapis | G | - | Extinct | Extinct | Lathyrus linifolius |
| Limenitis populi | F | Extinct | Extinct | - | Populus tremula |
| Lycaena hippothoe | G | Extinct | Extinct | Decrease | Rumex acetosa, Rumex acetosella |
| Lycaena virgaureae | G | Extinct | Extinct | Decrease | Rumex acetosa |
| Maculinea arion | G | - | Extinct | Extinct | Thymus, Origanum vulgare |
| Melitaea athalia | G | Extinct | Decrease | Decrease | Melampyrum |
| Melitaea cinxia | G | Extinct | - | Extinct | Veronica spicata, Plantago lanceolata |
| Nymphalis polychloros | F | Extinct | Extinct | - | Ulmus, Salix |
| Papilio machaon | G | Extinct | Extinct | Extinct | Peucedanum palustre, Pimpinella saxifraga |
| Plebejus optilete | W | Extinct | Decrease | Unchanged | Andromeda polifolia, Vaccinium oxycoccos, Vaccinium uliginosum |
| Polyommatus semiargus | G | Extinct | Extinct | Decrease | Trifolium pratense |
| Pyrgus malvae | G | Unchanged | Decrease | Decrease | Fragaria vesca, Potentilla |
| Satyrium pruni | G | Extinct | - | Decrease | Prunus spinosa |
| Thecla betulae | G | Extinct | - | Extinct | Prunus spinosa, Prunus padus |
| Zygaena filipendulae* | G | Unchanged | Extinct | Decrease | Lotus corniculatus |
| Zygaena lonicerae* | G | Extinct | Extinct | Decrease | Trifolium medium |
| Zygaena viciae* | G | Extinct | Unchanged | Decrease | Lathyrus linifolius, Vicia cracca |
After these two pioneering papers, long term declines in countryside butterfly diversity have been documented in detailed studies from the focal area of the present report (Table 1). From Kullaberg in north-western Skåne, a 45% loss from the initial 50 butterfly species has been documented between 1953 and 2005 within a 1000 hectare area of mixed forest and agricultural land (
It is worth noticing that most historical Swedish butterfly information on trends is based on presence/absence data as relatively few quantitative analyses have been carried out over time (
In total, 130 butterflies and burnet moths have been recorded in Sweden; 117 of these are resident and another 13 species occur sporadically. While no species have become extinct from Sweden during the last four decades, 40 butterfly and burnet moth species are included in the latest Red List (
Butterfly and burnet moth species listed in the Swedish Red List as extinct from the studied counties in southern Sweden (
| Species | Skåne | Blekinge | Gotland | Öland | Kalmar | Kronoberg | Jönköping | Halland | Västergötland | Östergötland |
|---|---|---|---|---|---|---|---|---|---|---|
| Argynnis niobe | † | † | ||||||||
| Coenonympha hero | † | † | ||||||||
| Euphydryas aurinia | † | † | † | |||||||
| Euphydryas maturna | † | † | ||||||||
| Hamearis lucina | † | † | ||||||||
| Limenitis camilla | † | |||||||||
| Lopinga achine | † | |||||||||
| Lycaena hippothoe | † | |||||||||
| Maculinea arion | † | † | † | † | ||||||
| Melitaea britomartis | † | † | ||||||||
| Melitaea cinxia | † | |||||||||
| Melitaea diamina | † | |||||||||
| Parnassius apollo | † | † | † | † | † | † | ||||
| Parnassius mnemosyne | † | † | † | † | † | |||||
| Polyommatus dorylas | † | † | ||||||||
| Pyrgus alveus | † | † | † | |||||||
| Satyrium ilicis | † | † | ||||||||
| Zygaena osterodensis | † | † | ||||||||
| Total | 10 | 5 | 2 | 2 | 2 | 4 | 5 | 4 | 4 | 4 |
In Sweden, we presently have 12 of the 17 different grassland species included in the European Butterfly Indicator for Grassland species (
Butterflies and burnet moths associated with semi-natural grasslands in the area: A Scarce Copper (Lycaena virgaureae) B Purple-edged Copper (Lycaena hippothoe) C Mazarine Blue (Polyommatus semiargus) D Amanda’s Blue (Polyommatus amandus) E New Forest Burnet (Zygaena viciae) and Narrow-bordered Five-spot Burnet (Zygaena lonicerae). Photos: Markus Franzén.
Among species associated with forest glades and wetlands, Moorland Clouded Yellow (Colias palaeno), Pearl-Bordered Fritillary (Boloria euphrosyne) and the glade-inhabiting Wood White (Leptidea sinapis) have all decreased markedly (
The land use in the focal area has changed dramatically over the last decades. Before Sweden joined the European Union in the early 1990s, there was a period when large farmland areas were used as set-asides in order to reduce subsidised production of wheat and other crops. The set-asides of these areas had a major positive impact on the population sizes of several bird species (
Three examples of sites less suited for butterflies: A Intensified hay cutting of ley fields has a strong negative effect on the butterfly fauna. Here is a storage place for bale silage close to a former Clouded Apollo, Parnassius mnemosyne, site in Blekinge B Succession of former semi-natural grasslands due to abandonment and C Intensive grazing early in the season as here by sheep can be devastating for many butterflies, their eggs, larvae and pupae as well as for nectar resources. Photos: Markus Franzén.
Another trend in recent years has been to harvest hay earlier and earlier, moving the onset of hay harvest back from around Midsummer to early June, and now often to late May (
Wetlands in the agricultural landscape have been drained in many places, and the butterflies of these areas were often seeking nectar on the surrounding meadows and pastures. Thus, because of the interactive effect of wetland draining and flower-rich pastures and meadows disappearing, species associated with wetlands have declined in the area during the last 50 years (
The present results from southern Sweden show an interesting parallel to studies elsewhere in Europe (
Current management and the European Union Common Agricultural Policy (CAP) regulations as applied in Sweden and elsewhere (
Based on the examples above, on conservation practitioners experience (cf.
- Later grazing, and grazing in segmented/split parcels so that some parts of pastures can grow taller and keep nectar resources longer in summer.
- Reduced grazing pressure in spring and early summer on herb-rich sites.
- Rotational grazing with some semi-natural grassland grazed only in late summer in some years.
- Hay cutting later in summer, preferably in late July, and once per season, followed by grazing of cattle in September–October.
- Herb rich grasslands should preferably be grazed by cattle or horses rather than sheep.
- If sheep are used, grazing needs careful management and timing to minimize negative effects on nectar resources.
- Implementation of policy measures that reduce in the overall nitrogen deposition from the atmosphere.
- Burning grasslands when the vegetation is dry in early spring, before the middle of April.
- Soil disturbance measures where fertilized top soil is removed, particularly at sun-exposed, sandy sites.
- Young invading trees (bushes), expanding shrubs, invading brackens and other expanding plant species should be removed.
- Spruce and pine plantations must cease on low fertility, semi-natural grassland sites.
- Implementation of compensation action from activities with negative impacts on butterfly habitats.
Markus Franzén was supported by the STEP-project (grant 244090–STEP–CP–FP;