Research Article |
|
Corresponding author: Orsolya Valkó ( valkoorsi@gmail.com ) Academic editor: Stefano Chelli
© 2020 Balázs Deák, Orsolya Valkó, Csaba Albert Tóth, Ágnes Botos, Tibor József Novák.
This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation:
Deák B, Valkó O, Tóth CA, Botos Á, Novák TJ (2020) Legacies of past land use challenge grassland recovery – An example from dry grasslands on ancient burial mounds. Nature Conservation 39: 113-132. https://doi.org/10.3897/natureconservation.39.52798
|
Abstract
Due to large-scale agricultural intensification, grasslands are often restricted to habitat islands in human-transformed landscapes. There are approximately half a million ancient burial mounds built by nomadic steppic tribes in the Eurasian steppe and forest steppe zones, which act as habitat islands for dry grassland vegetation. Land use intensification, such as arable farming and afforestation by non-native woody species are amongst the major threats for Eurasian dry grasslands, including grasslands on mounds. After the launch of the Good Agricultural and Environmental Condition framework of the European Union, in Hungary there is a tendency for ceasing crop production and cutting non-native woody plantations, in order to conserve these unique landmarks and restore the historical grassland vegetation on the mounds. In this study, restoration prospects of dry grassland habitats were studied on kurgans formerly covered by croplands and Robinia pseudoacacia plantations. Soil and vegetation characteristics were studied in thespontaneously recovering grasslands. The following questions were addressed: 1; How does site history affect the spontaneous grassland recovery? 2; Do residual soil nutrients play a role in grassland recovery? In former croplands, excess phosphorus, while in former Robinia plantations, excess nitrogen was present in the soil even four years after the land use change and grassland vegetation was in an early or mid-successional stage both on the mounds. The results showed that, without proper management measures, recovery of grassland vegetation is slow on mounds formerly used as cropland or black locust plantation. However, restoration efforts, focused on the restoration of mounds formerly covered by croplands, can be more effective compared to the restoration of mounds formerly covered by forest plantations.
Keywords
cropland, grassland restoration, kurgan, nitrogen, phosphorus, Robinia pseudoacacia, soil, steppe
Introduction
In intensively used agricultural landscapes, the remaining natural and semi-natural habitats often occur on small natural features (SNFs). Many of SNFs, such as verges, field margins and midfield islets are physically inappropriate for agricultural utilisation (
The Cross-Compliance system of the European Union Common Agricultural Policy is a progressive initiative that contributes to a more environmental-friendly agriculture. One of its pillars is the Good Agricultural and Environmental Condition framework, which contributes to the development of a long-term and ecologically sustainable agricultural environment (
In grassland restoration, spontaneous recovery became increasingly acknowledged (
Here we study two scenarios of grassland recovery: recovery on former croplands and former plantations on mounds. Ploughing and afforestation are responsible for the reduction of grassland area and decline of grassland species richness in many parts of Eurasia (
The objective was to evaluate the prospects for restoring grasslands in degraded (ploughed and planted with black locust) ancient burial mounds, in order to provide information that will support the development of management plans, restoration and conservation of the local biota. We asked the following questions: 1; How does site history affect the spontaneous grassland recovery? 2; Do residual soil nutrients play a role in grassland recovery? Our final goal was to give recommendations for the restoration of grasslands on variously degraded burial mounds.
Material and methods
Study sites
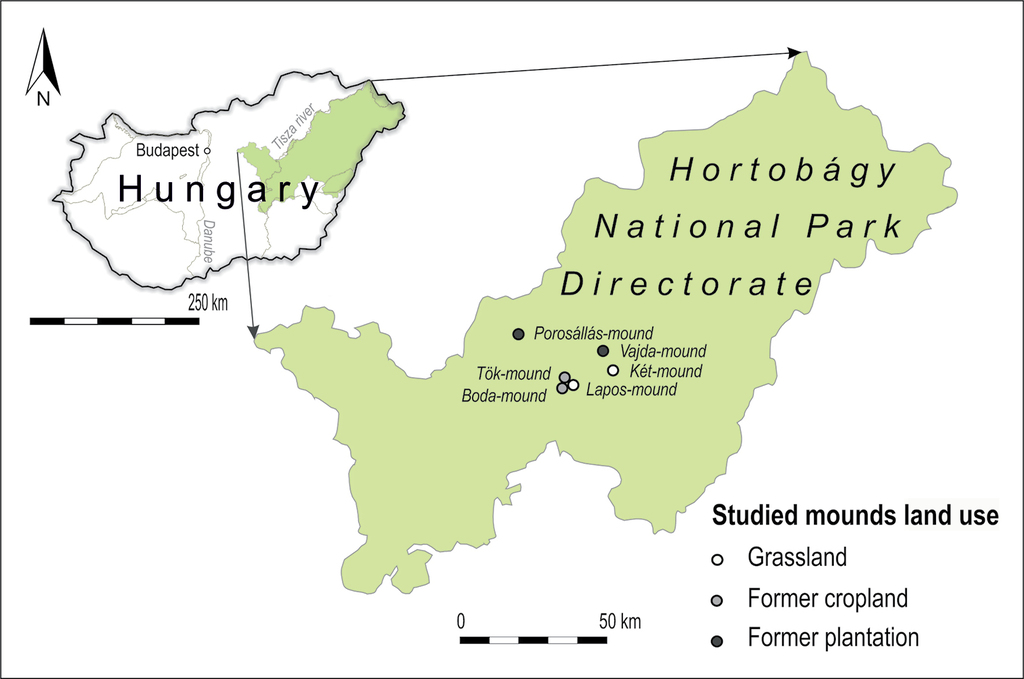
We studied soil and vegetation of six mounds situated in the Hortobágy National Park, EastHungary (Figure
The vegetation of four surveyed mounds was formerly seriously degraded: two mounds (Porosállás- and Vajda-mounds) were formerly covered by black locust (R. pseudoacacia) plantations and two mounds were used as arable fields (Boda- and Tök-mounds). According to the oldest available orthophotos, all the two mounds with former croplands were already ploughed and the two mounds with Robinia plantations were already afforested in 1961. Based on archive descriptions, we can estimate that afforestation lasted for at least 80 years and crop production for at least 200 years on the studied mounds. Spontaneous grassland recovery started in 2012 in all sites: plantations have been cut and ploughing was stopped. As reference, we selected two mounds (Kettős- and Lapos-mounds) with well-preserved pristine grassland vegetation. Figure
Field sampling
Soil conditions were sampled in ten randomly distributed 1 m × 1 m permanent plots on each mound. From each plot, three subsamples were collected with 100 cm3 stainless steel sampling cylinders, representing the uppermost 5 cm of the soil. First soil sampling was carried out in late June 2014 and it was repeated in late June 2016. On each mound, we designated ten 1 m × 1 m plots (altogether 60 plots), in which we recorded the percentage cover of vascular plant species in June 2016.
Laboratory analyses
Soil subsamples from the same plots and same year were then mixed and, after homogenisation, dried at 40 °C until weight constancy (approximately three days). In the laboratory, pH (H2O; KCl), plant available nitrogen content and plant available phosphorus content were measured. Soil pH was measured with standard glass electrode in 1:2.5 suspensions prepared with water (pHH2O), and KCl solution (pHKCl), respectively (MSZ-08-0206:1978 2.1). Plant available P was determined after extraction with 0.1 M ammonium-lactate solution buffered with 0.4 M acetic acid at pH 3.7. Plant available NO3--N was extracted with 1 M KCl solution. Both N and P content of extracts were determined by spectrophotometric measurements, according the Hungarian standards (MSZ 20135 1999).
Data processing
We calculated cover-weighted scores of Ellenberg ecological indicator values for water (WB), nutrient (NB) and light (LB) adapted to the Hungarian conditions (
For visualising the vegetation patterns on the mounds with different site history, we used Detrended Correspondence Analysis (DCA), based on specific cover scores. Soil nitrate and phosphorus content were included as overlay (CANOCO 5;
To explore the effects of ‘site history’, time since grassland recovery started (‘year’) and their interaction (explanatory variables) on soil pH (pH(H2O); pH(KCl)), soil nitrate and phosphorus content (dependent variables), we used Repeated Measures General Linear Models (RM GLMs) accounting for the normal distribution of the dependent variables (
We used Generalised Linear Mixed Models (GLMMs) to explore the effects of ‘site history’ and two soil parameters (‘soil nitrate content’ and ‘soil phosphorus content’) (explanatory variables) on the naturalness index, ecological indicator values and the species richness and cover of grassland species and weeds in 2016 (dependent variables). The ID of the ‘study sites’ was included in the models as a random factor. Scores of naturalness index, ecological indicator values and cover of grassland and weed species were log-transformed to approximate them to normal distribution. Species number of grassland specialists and weeds were fitted using Poisson distribution with a loglink. We used Tukey’s test for calculating post-hoc pair-wise comparisons. The RM GLMs and GLMMs were calculated using IBM SPSS Statistics v. 22 programme (Armonk, NY: IBM Corp). Significance level was set at p ≤ 0.05.
Results
Soil characteristics
We did not detect any difference in the soil pH(H2O) and pH(KCl) of the studied mounds (Table
Effects of site history, year and their interaction on soil attributes (RM GLM). Notations: *** p < 0.001; ** p < 0.01; * p < 0.05; n.s.: non-significant.
| Parameter | Site history | Year | Site history × Year | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| dfnum | dfden | F | p | dfnum | dfden | F | p | dfnum | dfden | F | p | |
| pH(H2O) | 2 | 1.067 | 2.636 | n.s. | 1 | 1 | 0.425 | n.s. | 2 | 1.247 | 1.426 | n.s. |
| pH(KCl) | 2 | 1.088 | 2.822 | n.s. | 1 | 1 | 1.080 | n.s. | 2 | 1.323 | 1.972 | n.s. |
| NO3--N content | 2 | 1.467 | 37.289 | *** | 1 | 1 | 2.190 | n.s. | 2 | 1.331 | 0.138 | n.s. |
| P content (P2O5) | 2 | 1.730 | 10.592 | ** | 1 | 1 | 17.590 | ** | 2 | 1.363 | 3.120 | n.s. |
Soil characteristics (A – pH(H2O), B – pH(KCl), C – NO3--N content and D – P content (P2O5)) measured in the studied mounds (grasslands, former croplands and former plantations). White boxes represent data from 2014, grey boxes represent data from 2016. Different letters indicate significant differences between groups (Tukey test, p ≤ 0.05).
Vegetation
We found altogether 92 vascular plant species on the studied mounds. Total species numbers were 64 in the grasslands, 50 in the former croplands and 24 in the former plantations. The vegetation composition of mounds with a different site history was well separated on the DCA ordination (Figure
DCA plot of the vegetation of study sites, based on the species composition. Soil nitrate and phosphorus content were included as an overlay. Notations: squares – grasslands; diamonds – former croplands, circles – former plantations. Eigenvalues were 0.753 and 0.548 for the first and second axis, respectively. Cumulative explained variance of the first and the second axis were 12.72% and 21.98%, respectively.
Indicator species of grasslands were Carex praecox Schreb., Koeleria cristata (L.) Pers., Salvia nemorosa L. and Festuca pseudovina Hack. ex Wiesb. (Table
Results of indicator species analyses of the vegetation of mounds with different site history. Notations: G – mounds covered by grassland; A – mounds formerly covered by arable land; P – mounds formerly covered by Robinia plantation; *** p < 0.001; ** p < 0.01; * p < 0.05.
| Species | Site history | Indicator value | p | Frequency |
|---|---|---|---|---|
| Carex praecox Schreb. | G | 0.55 | *** | 11 |
| Koeleria cristata (L.) Pers. em. Borbás ex Domin | G | 0.49 | *** | 14 |
| Salvia nemorosa L. | G | 0.40 | *** | 8 |
| Festuca pseudovina Hack. ex Wiesb. | G | 0.40 | ** | 13 |
| Elymus hispidus (Opiz) Melderis | G | 0.35 | ** | 7 |
| Arenaria serpyllifolia L. | G | 0.33 | * | 18 |
| Plantago lanceolata L. | G | 0.32 | ** | 8 |
| Bromus hordeaceus L. | G | 0.32 | * | 14 |
| Trifolium retusum L. | G | 0.30 | ** | 6 |
| Euphorbia virgata Waldst. et Kit. | G | 0.30 | *** | 6 |
| Erodium cicutarium (L.) L’Hér. | G | 0.26 | * | 8 |
| Eryngium campestre L. | G | 0.25 | * | 5 |
| Trifolium striatum L. | G | 0.25 | ** | 5 |
| Stipa capillata L. | G | 0.20 | * | 4 |
| Cerastium semidecandrum L. | G | 0.20 | * | 4 |
| Bromus tectorum L. | A | 0.84 | *** | 18 |
| Torilis arvensis (Huds.) Link | A | 0.49 | *** | 12 |
| Convolvulus arvensis L. | A | 0.44 | * | 34 |
| Alopecurus pratensis L. | A | 0.43 | *** | 12 |
| Achillea collina Becker ex Rchb. | A | 0.42 | ** | 18 |
| Veronica arvensis Murray | A | 0.34 | ** | 8 |
| Geranium molle L. | A | 0.26 | * | 9 |
| Xanthium strumarium L. | A | 0.25 | ** | 5 |
| Vicia grandiflora Scop. | A | 0.20 | * | 4 |
| Lolium perenne L. | A | 0.20 | * | 4 |
| Elymus repens (L.) Gould | P | 0.65 | *** | 30 |
| Ballota nigra L. | P | 0.51 | *** | 14 |
| Bromus sterilis L. | P | 0.35 | *** | 7 |
| Silene alba (Mill.) E.H.L. Krause | P | 0.28 | * | 11 |
| Galium aparine L. | P | 0.26 | * | 13 |
| Conium maculatum L. | P | 0.20 | * | 4 |
Site history significantly affected the naturalness of the vegetation, it was the lowest in former plantations and the highest in grasslands (Table
Effects of site history, soil nitrate and phosphorus content on the vegetation characteristics of the studied mounds (Generalised Linear Mixed Models). *** p < 0.001; ** p < 0.01; * p < 0.05; n.s., non-significant. Notations: NB, WB, LB: cover-weighted means of ecological indicator values for nutrients, water and light, respectively.
| Site history | Nitrate | Phosphorus | ||||
| F | p | F | p | F | p | |
| Naturalness | 46.35 | *** | 5.15 | * | 5.40 | * |
| Species richness | ||||||
| Grassland species | 17.59 | *** | 0.19 | n.s. | 1.80 | n.s. |
| Weeds | 1.95 | n.s. | 0.61 | n.s. | 0.61 | n.s. |
| Cover | ||||||
| Grassland species | 17.74 | *** | 0.20 | n.s. | 3.09 | n.s. |
| Weeds | 70.27 | *** | 0.03 | n.s. | 0.01 | n.s. |
| Ecological indicator values | ||||||
| NB | 9.02 | *** | 0.93 | n.s. | 1.19 | n.s. |
| WB | 7.97 | *** | 2.51 | n.s. | 0.44 | n.s. |
| LB | 21.38 | *** | 13.09 | *** | 3.25 | n.s. |
Vegetation characteristics in the studied mounds (grasslands, former croplands and former plantations): A species richness of grassland species B species richness of weeds C cover of grassland species D cover of weeds E cover-weighted ecological indicator scores for nutrients (NB) F cover-weighted ecological indicator scores for water (WB) G cover-weighted ecological indicator scores for light (LB) H naturalness score. Different letters indicate significant differences between groups (Tukey test, p ≤ 0.05).
Discussion
Soil characteristics
We found that pH was highest, close to 7 or even greater, in former cropland in 2014. The likely reason for this is that, in croplands, there was no organic matter accumulation on the surface and the surficial soil layer was constantly mixed with subsoil and the subsoils’ carbonate saturation status was always higher in these soils than at the surface. During the study period, pH was not changed significantly in any of the mounds, but a slight increase in former plantation and slight decrease on former cropland couldbe detected. Soil pH was much more heterogeneous in the grasslands compared to the former croplands and plantations. This supports the findings of the
Plant available N content was the lowest in the former cropland and its amount did not change by time since abandonment. This is due to the additive effect of the depletion of soil N stocks by decades-long cultivation and the slow soil N-recovery during secondary succession (
Plant available P content was the highest in the former croplands in 2014 as result of former P-fertilisation, but after the abandonment, it started to decrease significantly. As P has a low mobility in soils, this decrease can be a result of the P consumption by the vegetation and soil microbes. In the grasslands and former plantations, P content was similarly low, in both cases low pH likely facilitated the mobilisation (
Vegetation changes
We found that, four years after land use change, grassland vegetation was in an early or mid-successional stage, both on the mounds formerly used as cropland and forest plantation. The three vegetation types were clearly separated by their species composition (Figure
Species composition of weeds reflected well the site history. Weeds are generally R-strategists and have a dense and persistent seed bank (
The reason for the low success of vegetation recovery on former plantations is the legacy of the former woody vegetation and forestry practices. For establishing a forest plantation, more drastic soil works are needed compared to arable use. Even though the soil works are not so frequent, such as in the case of arable use, they can affect the soil structure in deeper layers. Thus, it affects the chemical properties of the soil in a more intense way; furthermore, deep ploughing transports the seed bank of grassland species to such deep layers (even 1 m deep) from where they are not able to germinate and re-establish. Due to the shading effect of woody vegetation, a milder micro-climate is present in the understorey of the woody habitats (
Conclusions
Our results suggest that the legacy of a former intensive land use (i.e. cropland and plantation) is more complex than the effect of excess soil nutrients. Even though former croplands were characterised by excess P and former plantations by excess N (Figure
We found that, without proper management measures, recovery of grassland vegetation is slow on mounds formerly used as cropland or black locust plantation. This is in line with the findings of
Even though spontaneous regeneration of grassland habitats can be a good solution in nature conservation practice, it might be risky as the vegetation development is often unpredictable. The outcome of spontaneous succession is highly dependent on initial site conditions, including land use intensity, landscape context, climaticand edaphic factors. Spontaneous recovery is often unpredictable also due to the founder effect, i.e. that the vegetation composition of late successional phases strongly depends on the initial plant assemblages (
Acknowledgements
The study was supported by the NKFI KH 130338, NKFI KH 126476, NKFI FK 124404 and NKFIH-1150-6/2019 grants. BD, OV and TJN were supported by the Bolyai János Scholarship of the Hungarian Academy of Sciences. TJN was supported by the ÚNKP-19-4-DE-129 New National Excellence Program (Bolyai+) of the Ministry for Innovation and Technology. The authors are grateful to Iva Apostolova and Igor Soares dos Santos for their useful suggestions on the manuscript.
References
- Albert Á-J, Kelemen A, Valkó O, Miglécz T, Csecserits A, Rédei T, Deák B, Tóthmérész B, Török P (2014) Secondary succession in sandy old-fields: A promising example of spontaneous grassland recovery. Applied Vegetation Science 17(2): 214–224. https://doi.org/10.1111/avsc.12068
- Alt F, Oelmann Y, Herold N, Schrumpf M, Wilcke W (2011) Phosphorus partitioning in grassland and forest soils of Germany as related to land‐use type, management intensity, and land use–related pH. Journal of Plant Nutrition and Soil Science 174(2): 195–209. https://doi.org/10.1002/jpln.201000142
- An H, Zhang B, Thomas BW, Beck R, Willms WD, Li Y, Hao X (2019) Short term recovery of vegetation and soil after abandoning cultivated mixed-grass prairies in Alberta, Canada. Catena 173: 321–329. https://doi.org/10.1016/j.catena.2018.10.017
- Auffret AG (2011) Can seed dispersal by human activity play a useful role for the conservation of European grasslands? Applied Vegetation Science 14(3): 291–303. https://doi.org/10.1111/j.1654-109X.2011.01124.x
- Bede Á, Csathó AI (2019) Complex characterization of kurgans in the Csanádi-hát region, Hungary. Tájökológiai Lapok 17(2): 131–145.
- Bede Á, Salisbury RB, Csathó AI, Czukor P, Páll DG, Szilágyi G, Sümegi P (2015) Report of the complex geoarcheological survey at the Ecse-halom kurgan in Hortobágy, Hungary. Central European Geology 58(3): 268–289. https://doi.org/10.1556/24.58.2015.3.5
- Biró M, Bölöni J, Molnár Z (2018) Use of long-term data to evaluate loss and endangerment status of Natura 2000 habitats and effects of protected areas. Conservation Biology 3(3): 660–671. https://doi.org/10.1111/cobi.13038
- Bolat I, Kara Ö, Sensoy H, Yüksel K (2015) Influences of Black Locust (Robinia pseudoacacia L.) afforestation on soil microbial biomass and activity. iForest – Biogeosciences and Forestry 9: 171–177. https://doi.org/10.3832/ifor1410-007
- Borhidi A (1995) Social behaviour types, the naturalness and relative ecological indicator values of the higher plants in the Hungarian Flora. Acta Botanica Hungarica 39: 97–181.
- Borhidi A, Kevey B, Lendvai G (2012) Plant communities of Hungary. Akadémiai Kiadó, Budapest.
- Bossuyt B, Honnay O (2008) Can the seed bank be used for ecological restoration? An overview of seed bank characteristics in European communities. Journal of Vegetation Science 19(6): 875–884. https://doi.org/10.3170/2008-8-18462
- Brady M, Kellermann K, Sahrbacher C, Jelinek L (2009) Impacts of decoupled agricultural support on farm structure, biodiversity and landscape mosaic: Some EU results. Journal of Agricultural Economics 60(3): 563–585. https://doi.org/10.1111/j.1477-9552.2009.00216.x
- Cierjacks A, Kowarik I, Joshi J, Hemplel S, Ristow M, von der Lippe M, Weber E (2013) Biological flora of the British Isles: Robinia pseudoacacia. Journal of Ecology 101(6): 1623–1640. https://doi.org/10.1111/1365-2745.12162
- Csecserits A, Rédei T (2001) Secondary succession on sandy old-fields in Hungary. Applied Vegetation Science 4(1): 63–74. https://doi.org/10.1111/j.1654-109X.2001.tb00235.x
- Csecserits A, Halassy M, Kröel-Dulay G, Rédei T, Szitár K, Szabó R (2011) Different regeneration success of sandy old-fields in the forest-steppe region of Hungary. Plant Biosystems 145: 715–729. https://doi.org/10.1080/11263504.2011.601340
- Deák B, Valkó O, Alexander C, Mücke W, Kania A, Tamás J, Heilmeier H (2014) Fine-scale vertical position as an indicator of vegetation in alkali grasslands – case study based on remotely sensed data. Flora 209(12): 693–697. https://doi.org/10.1016/j.flora.2014.09.005
- Deák B, Valkó O, Török P, Kelemen A, Miglécz T, Szabó S, Szabó G, Tóthmérész B (2015) Micro-topographic heterogeneity increases plant diversity in old stages of restored grasslands. Basic and Applied Ecology 16(4): 291–299. https://doi.org/10.1016/j.baae.2015.02.008
- Deák B, Tóthmérész B, Valkó O, Sudnik-Wójcikowska B, Bragina TM, Moysiyenko II, Bragina TM, Apostolova I, Dembicz I, Bykov NI, Török P (2016a) Cultural monuments and nature conservation: The role of kurgans in maintaining steppe vegetation. Biodiversity and Conservation 25: 2473–2490. https://doi.org/10.1007/s10531-016-1081-2
- Deák B, Valkó O, Török P, Tóthmérész B (2016b) Factors threatening grassland specialist plants – A multi-proxy study on the vegetation of isolated grasslands. Biological Conservation 204: 255–262. https://doi.org/10.1016/j.biocon.2016.10.023
- Deák B, Tóth CA, Bede Á, Apostolova I, Bragina TM, Báthori F, Bán M (2019) Eurasian Kurgan Database – a citizen science tool for conserving grasslands on historical sites. Hacquetia 18(2): 185–193. https://doi.org/10.2478/hacq-2019-0007
- Deák B, Valkó O, Nagy DD, Török P, Torma A, Lőrinczi G, Kelemen A, Nagy A, Bede Á, Mizser S, Csathó AI, Tóthmérész B (2020) Habitat islands outside nature reserves – threatened biodiversity hotspots of grassland specialist plant and arthropod species. Biological Conservation 241: 108254. https://doi.org/10.1016/j.biocon.2019.108254
- Dembicz I, Moysiyenko II, Shaposhnikova A, Vynokurov D, Kozub L, Sudnik-Wójcikowska B (2016) Isolation and patch size drive specialist plant species density within steppe islands: A case study of kurgans in southern Ukraine. Biodiversity and Conservation 25(12): 2289–2307. https://doi.org/10.1007/s10531-016-1077-y
- Dembicz I, Szczeparska L, Moysiyenko II, Wódkiewicz M (2018) High genetic diversity in fragmented Iris pumila L. populations in Ukrainian steppe enclaves. Basic and Applied Ecology 28: 37–47. https://doi.org/10.1016/j.baae.2018.02.009
- Dudley N, Higgins-Zogib L, Mansourian S (2009) The links between protected areas, faiths, and sacred natural sites. Conservation Biology 23(3): 568–577. https://doi.org/10.1111/j.1523-1739.2009.01201.x
- Dufrêne M, Legendre P (1997) Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecological Monographs 67: 345–366. https://doi.org/10.2307/2963459
- Fekete R, Löki V, Urgyán R, Süveges K, Lovas-Kiss Á, Vincze O, Molnár VA (2019) Roadside verges and cemeteries: Comparative analysis of anthropogenic orchid habitats in the Eastern Mediterranean. Ecology and Evolution 9(11): 6655–6664. https://doi.org/10.1002/ece3.5245
- Godó L, Valkó O, Tóthmérész B, Török P, Kelemen A, Deák B (2017) Scale-dependent effects of grazing on the species richness of alkaline and sand grasslands. Tuexenia 37: 229–246.
- Grime JP (1998) Benefits of plant diversity to ecosystems: Immediate, filter and founder effects. Journal of Ecology 86(6): 902–910. https://doi.org/10.1046/j.1365-2745.1998.00306.x
- Hedberg P, Kotowski W (2010) New nature by sowing? The current state of species introduction in grassland restoration, and the road ahead. Journal for Nature Conservation 18(4): 304–308. https://doi.org/10.1016/j.jnc.2010.01.003
- Jacquemyn H, Roldán-Ruiz I, Honnay O (2010) Evidence for demographic bottlenecks and limited gene flow leading to low genetic diversity in a rare thistle. Conservation Genetics 11(5): 1979–1987. https://doi.org/10.1007/s10592-010-0089-5
- Jakobsson S, Bernes C, Bullock JM, Verheyen K, Lindborg R (2018) How does roadside vegetation management affect the diversity of vascular plants and invertebrates? A systematic review. Environmental Evidence 7(1): 17. https://doi.org/10.1186/s13750-018-0129-z
- Jentsch A, Beyschlag W (2003) Vegetation ecology of dry acidic grasslands in the lowland area of Central Europe. Flora 198(1): 3–25. https://doi.org/10.1078/0367-2530-00071
- Jírová A, Klaudisová A, Prach K (2012) Spontaneous restoration of target vegetation in old-fields in a central European landscape: A repeated analysis after three decades. Applied Vegetation Science 15(2): 245–252. https://doi.org/10.1111/j.1654-109X.2011.01165.x
- Kiehl K, Kirmer A, Donath T, Rasran L, Hölzel N (2010) Species introduction in restoration projects. Evaluation of different techniques for the establishment of semi natural grasslands in Central and Northwestern Europe. Basic and Applied Ecology 11(4): 285–299. https://doi.org/10.1016/j.baae.2009.12.004
- Knops JM, Tilman D (2000) Dynamics of soil nitrogen and carbon accumulation for 61 years after agricultural abandonment. Ecology 81(1): 88–98. https://doi.org/10.1890/0012-9658(2000)081[0088:DOSNAC]2.0.CO;2
- Kottek M, Grieser J, Beck C, Rudolf B, Rubel F (2006) World Map of Köppen-Geiger Climate Classification updated. Meteorologische Zeitschrift 15(3): 259–263. https://doi.org/10.1127/0941-2948/2006/0130
- Lakatos M, Szentimrey T, Bihari Z, Szalai S (2013) Creation of a homogenized climate database for the Carpathian region by applying the MASH procedure and the preliminary analysis of the data. Idöjárás 117: 143–158.
- Lencová K, Prach K (2011) Restoration of hay meadows on ex-arable land: Commercial seed mixtures vs. spontaneous succession. Grass and Forage Science 66(2): 265–271. https://doi.org/10.1111/j.1365-2494.2011.00786.x
- Lindborg R, Plue J, Andersson K, Cousins SAO (2014) Function of small habitat elements for enhancing plant diversity in different agricultural landscapes. Biological Conservation 169: 206–213. https://doi.org/10.1016/j.biocon.2013.11.015
- Lisetskii FN, Sudnik-Wójcikowska B, Moysiyenko II (2016) Flora differentiation among local ecotypes in the transzonal study of forest-steppe and steppe mounds. The Biological Bulletin 43(2): 169–176. https://doi.org/10.1134/S1062359016010106
- Löki V, Molnár VA, Süveges K, Heimeier H, Takács A, Nagy T, Fekete R, Lovas-Kiss Á, Kreutz CAJK, Sramkó G, Tökölyi J (2019) Predictors of conservation value of Turkish cemeteries: A case study using orchids. Landscape and Urban Planning 186: 36–44. https://doi.org/10.1016/j.landurbplan.2019.02.016
- Matamala R, Jastrow JD, Miller RM, Garten CT (2008) Temporal changes in C and N stocks of restored prairie: Implications for C sequestration strategies. Ecological Applications 18(6): 1470–1488. https://doi.org/10.1890/07-1609.1
- Matus G, Tóthmérész B, Papp M (2003) Restoration prospects of abandoned species-rich sandy grassland in Hungary. Applied Vegetation Science 6(2): 169–178. https://doi.org/10.1111/j.1654-109X.2003.tb00577.x
- Melnik K, Landhäusser SM, Devito K (2017) Role of microtopography in the expression of soil propagule banks on reclamation sites. Restoration Ecology 26(S2): S200–S210. https://doi.org/10.1111/rec.12587
- Moeslund JE, Arge L, Bøcher PK, Dalgaard T, Odgaard MV, Nygaard B, Svenning JC (2013) Topographically controlled soil moisture is the primary driver of local vegetation patterns across a lowland region. Ecosphere 4(7): 1–26. https://doi.org/10.1890/ES13-00134.1
- Molnár VA, Nagy T, Löki V, Süveges K, Takács A, Bódis J, Tökölyi J (2017) Turkish graveyards as refuges for orchids against tuber harvest. Ecology and Evolution 7(24): 11257–11264. https://doi.org/10.1002/ece3.3562
- Penksza K, Loksa G, Barczi A, Joó K, Malatinszky Á (2011) Effects of extrazonal and climatic conditions on the vegetation of kurgans. A pilot study from the Hortobágy (Csípő-halom). In: Pető Á, Barczi A (Eds) Kurgan studies: An environmental and archaeological multiproxy study of burial mounds in the Eurasian steppe zone. BAR International Series, Oxford, 347–350.
- Prach K, Pyšek P (2001) Using spontaneous succession for restoration of human-disturbed habitats: Experience from Central Europe. Ecological Engineering 17(1): 55–62. https://doi.org/10.1016/S0925-8574(00)00132-4
- Prach K, Řehounková K (2008) Spontaneous vegetation succession in gravel–sand pits: A potential for restoration. Restoration Ecology 16(2): 305–312. https://doi.org/10.1111/j.1526-100X.2007.00316.x
- R Core Team (2020) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna.
- Rákóczi A, Barczi A (2014) Protected landscape elements in the European Union, the influence of EC decree 73/2009 on the condition of Hungarian mounds. Tájökológiai Lapok 12(1): 95–105. [Védett tájelemek az Európai Unióban, a 73/2009 EK rendelet hatásai a magyar kunhalmok állapotára]
- Roberts DW (2019) Ordination and Multivariate Analysis for Ecology. Version 2.0-1 URL http://ecology.msu.montana.edu/labdsv/R
- Ruprecht E (2005) Secondary succession in old-fields in the Transylvanian Lowland (Romania). Preslia 77: 145–157.
- Ruprecht E (2006) Successfully recovered grassland: A promising example from Romanian old-fields. Restoration Ecology 14(3): 473–480. https://doi.org/10.1111/j.1526-100X.2006.00155.x
- Sudnik-Wójcikowska B, Moysiyenko II, Zachwatowicz M, Jabłońska E (2011) The value and need for protection of kurgan flora in the anthropogenic landscape of steppe zone in Ukraine. Plant Biosystems 145(3): 638–653. https://doi.org/10.1080/11263504.2011.601335
- Ter Braak C, Šmilauer P (2012) Canoco Reference Manual and User’s Guide: Software for Canonical Community Ordination (version 5.0). Microcomputer Power, Ithaca, NY, USA, 496 pp.
- Tölgyesi C, Török P, Hábenczyus AA, Bátori Z, Valkó O, Deák B, Tóthmérész B, Erdős L, Kelemen A (2020) Underground deserts below fertility islands? – Woody species desiccate lower soil layers in sandy drylands. Ecography. https://doi.org/10.1111/ecog.04906
- Török P, Vida E, Deák B, Lengyel S, Tóthmérész B (2011a) Grassland restoration on former croplands in Europe: An assessment of applicability of techniques and costs. Biodiversity and Conservation 20(11): 2311–2332. https://doi.org/10.1007/s10531-011-9992-4
- Török P, Kelemen A, Valkó O, Deák B, Lukács BA, Tóthmérész B (2011b) Lucerne dominated fields recover native grass diversity without intensive management actions. Journal of Applied Ecology 48(1): 257–264. https://doi.org/10.1111/j.1365-2664.2010.01903.x
- Török P, Miglécz T, Valkó O, Kelemen A, Deák B, Lengyel S, Tóthmérész B (2012) Recovery of native grass biodiversity by sowing on former croplands: Is weed suppression a feasible goal for grassland restoration? Journal for Nature Conservation 20(1): 41–48. https://doi.org/10.1016/j.jnc.2011.07.006
- Tóth CA (2006) Results of the national mound cadastering from the aspect of geological conservation. Acta GGM Debrecina Geology, Geomorphology, Physical Geography Series 1: 129–135.
- Tóth CA, Rákóczi A, Tóth S (2018) Protection of the state of prehistoric mounds in Hungary: Law as a conservation measure. Conservation and Management of Archaeological Sites 20(3): 113–142. https://doi.org/10.1080/13505033.2018.1486125
- Tóth CA, Deák B, Nyilas I, Bertalan L, Valkó O, Novák T (2019) Iron age burial mounds as refugia for steppe specialist plants and invertebrates – case study from the Zsolca mounds (NE Hungary). Hacquetia 18(2): 195–206. https://doi.org/10.2478/hacq-2019-0009
- Valkó O, Török P, Tóthmérész B, Matus G (2011) Restoration potential in seed banks of acidic fen and dry-mesophilous meadows: Can restoration be based on local seed banks? Restoration Ecology 19(101): 9–15. https://doi.org/10.1111/j.1526-100X.2010.00679.x
- Valkó O, Tóth K, Kelemen A, Miglécz T, Sonkoly J, Tóthmérész B, Török P, Deák B (2018) Cultural heritage and biodiversity conservation – Plant introduction and practical restoration on ancient burial mounds. Nature Conservation 24: 65–80. https://doi.org/10.3897/natureconservation.24.20019
- Vítková M, Müllerová J, Sádlo J, Pergl J, Pyšek P (2017) Black locust (Robinia pseudoacacia) beloved and despised: A story of an invasive tree in Central Europe. Forest Ecology and Management 384: 287–302. https://doi.org/10.1016/j.foreco.2016.10.057
- Zuur AF, Ieno EN, Walker N, Saveliev AA, Smith GM (2009) Mixed Effects Models and Extensions in Ecology. Springer, New York, 574 pp. https://doi.org/10.1007/978-0-387-87458-6